Diabetes Mellitus in a nutshell
How does Diabetes Mellitus develop?
Type I DM may be caused by an immune mediated process or idiopathic (occurring without known cause).
The pancreas has structures called islets of Langerhans which are made up of beta cells that produce insulin. Insulin is a hormone that helps to burn and store sugar in the body. Insulin functions like a key; it "opens" the cells' door so glucose can enter the cells. Insulin helps to transport glucose across cell membranes (Potts & Mandleco, 2007; TImby & Smith, 2013). Since the beta cells in the
pancreas are destroyed by the autoimmune response, they do not produce these crucial hormones, therefore sugar will accumulate in the bloodstream and spill into the urine. Since the cells do not have glucose to derive energy from, the cells start to breakdown fat (lipolysis) and protein (proteolysis) from the body in order to supply the child's energy needs. This in turn causes the patient to lose weight and slows down growth in the child (Klossner & Hatfield, 2010). With the impaired production of insulin, the liver increases its production of glycogen which then converts into glucose (glycogenesis) in order to meet the body's needs. Hyperlipidemia and ketone formation occur with this process (Potts & Mandleco, 2007). Ketone formation occurs from breakdown of fats, leading to
ketonuria and ketonemia (ketones in the urine and in the blood, respectively). When the glucose levels in the blood exceed
150-180 mg/dL, glucose is filtered out of the serum and into the urine due to renal threshold limits (Potts & Mandleco, 2007). When this occurs, polyuria ensues due to an osmotic shift and additional excretion of water from glucosuria. As a result of glucosuria and additional fluid loss, the thirst center is activated and polydipsia, extreme thirst, develops. In addition, the hunger center is timulated as less glucose is used for energy production and storage, this is called polyphagia (excessive hunger) (Potts & Mandleco, 2007).
As hyperglycemia ensues and persists, the symptoms become more severe: fluid loss becomes severe, oral consumption does not replenish losses, dehydration occurs, weight loss continues, and electrolyte imbalances result from the osmotic diuresis and cell death (Potts & Mandleco, 2007).
Ketones, acid bodies, are initially buffered by stores of bicarbonate (Potter & Mandleco, 2007). However, when the kidney eventually loses its ability to buffer these ketone bodies, and the bicarbonate stores are depleted, metabolic acidosis worsens (Potts & andleco, 2007). Metabolic acidosis activates respiratory compensation. This is initially seen when patients develop increased respirations, known as Kussmaul respirations characterized by deep, rapid, labored breathing (hyperventilation). Flushed cheeks and an acetone odor (with a fruity smell) on the breath similar to nail polish remover or rotten apples, are also noticed ( Potts & Mandleco, 2007) along with drowsiness, dry skin, cherry-red lips, nausea and vomiting (Klossner & Hatfield, 2010). These symptoms are known as Diabetic Ketoacidosis (DKA). If untreated, the patient may lapse into a coma state and "exhibit dehydration, electrolyte imbalance, rapid pulse, and subnormal temperature and blood pressure" (Klossner & Hatfield, 2010, p. 855). DKA can lead to death
when untreated. It causes body systems to shut down: circulatory collapse, renal shutdown, and consequently death.
The symptoms of DKA develop more suddenly in Type I diabetes due to the total cessation of insulin production (Timby & Smith, 2013). DKA is more common in Type I because they no longer produce insulin
Recent research by Dr. D. Faustmann and associates (2008) indicates that there is a genetic mutation in Type I DM that causes killer T-cell lymphocytes (which help to attack foreign invading organisms in our body) to attack and destroy the insulin-producing islet cells (p. 784). The hypothesis states that people with Type I may lack a protein marker that allows the T cells to identify natural cells as "native" or "self". So without this marker, the T cells mistake the islet cells as foreign and start attacking them. Research has found a way to destroy the T-cells and regenerate new islet cells with bacillus Calmette-Guerin (BCG) vaccine, a vaccine typically given for tuberculosis. For more information on this research, click here. This research could potentially cure Type I DM and allow for many other autoimmune disorderes to be cured as well.
Type I DM may be caused by an immune mediated process or idiopathic (occurring without known cause).
The pancreas has structures called islets of Langerhans which are made up of beta cells that produce insulin. Insulin is a hormone that helps to burn and store sugar in the body. Insulin functions like a key; it "opens" the cells' door so glucose can enter the cells. Insulin helps to transport glucose across cell membranes (Potts & Mandleco, 2007; TImby & Smith, 2013). Since the beta cells in the
pancreas are destroyed by the autoimmune response, they do not produce these crucial hormones, therefore sugar will accumulate in the bloodstream and spill into the urine. Since the cells do not have glucose to derive energy from, the cells start to breakdown fat (lipolysis) and protein (proteolysis) from the body in order to supply the child's energy needs. This in turn causes the patient to lose weight and slows down growth in the child (Klossner & Hatfield, 2010). With the impaired production of insulin, the liver increases its production of glycogen which then converts into glucose (glycogenesis) in order to meet the body's needs. Hyperlipidemia and ketone formation occur with this process (Potts & Mandleco, 2007). Ketone formation occurs from breakdown of fats, leading to
ketonuria and ketonemia (ketones in the urine and in the blood, respectively). When the glucose levels in the blood exceed
150-180 mg/dL, glucose is filtered out of the serum and into the urine due to renal threshold limits (Potts & Mandleco, 2007). When this occurs, polyuria ensues due to an osmotic shift and additional excretion of water from glucosuria. As a result of glucosuria and additional fluid loss, the thirst center is activated and polydipsia, extreme thirst, develops. In addition, the hunger center is timulated as less glucose is used for energy production and storage, this is called polyphagia (excessive hunger) (Potts & Mandleco, 2007).
As hyperglycemia ensues and persists, the symptoms become more severe: fluid loss becomes severe, oral consumption does not replenish losses, dehydration occurs, weight loss continues, and electrolyte imbalances result from the osmotic diuresis and cell death (Potts & Mandleco, 2007).
Ketones, acid bodies, are initially buffered by stores of bicarbonate (Potter & Mandleco, 2007). However, when the kidney eventually loses its ability to buffer these ketone bodies, and the bicarbonate stores are depleted, metabolic acidosis worsens (Potts & andleco, 2007). Metabolic acidosis activates respiratory compensation. This is initially seen when patients develop increased respirations, known as Kussmaul respirations characterized by deep, rapid, labored breathing (hyperventilation). Flushed cheeks and an acetone odor (with a fruity smell) on the breath similar to nail polish remover or rotten apples, are also noticed ( Potts & Mandleco, 2007) along with drowsiness, dry skin, cherry-red lips, nausea and vomiting (Klossner & Hatfield, 2010). These symptoms are known as Diabetic Ketoacidosis (DKA). If untreated, the patient may lapse into a coma state and "exhibit dehydration, electrolyte imbalance, rapid pulse, and subnormal temperature and blood pressure" (Klossner & Hatfield, 2010, p. 855). DKA can lead to death
when untreated. It causes body systems to shut down: circulatory collapse, renal shutdown, and consequently death.
The symptoms of DKA develop more suddenly in Type I diabetes due to the total cessation of insulin production (Timby & Smith, 2013). DKA is more common in Type I because they no longer produce insulin
Recent research by Dr. D. Faustmann and associates (2008) indicates that there is a genetic mutation in Type I DM that causes killer T-cell lymphocytes (which help to attack foreign invading organisms in our body) to attack and destroy the insulin-producing islet cells (p. 784). The hypothesis states that people with Type I may lack a protein marker that allows the T cells to identify natural cells as "native" or "self". So without this marker, the T cells mistake the islet cells as foreign and start attacking them. Research has found a way to destroy the T-cells and regenerate new islet cells with bacillus Calmette-Guerin (BCG) vaccine, a vaccine typically given for tuberculosis. For more information on this research, click here. This research could potentially cure Type I DM and allow for many other autoimmune disorderes to be cured as well.
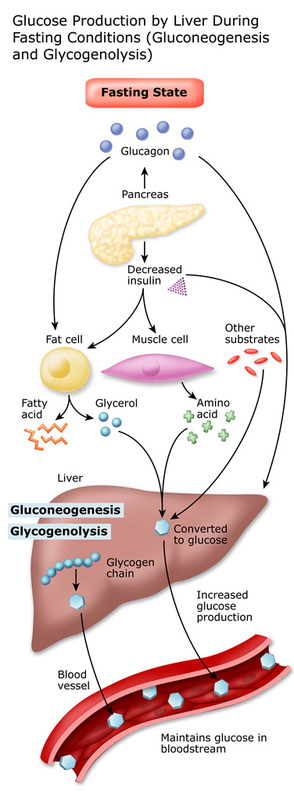
Type II DM on the other hand is more of an acquired and often times hereditary disorder. There appears to be no specific gene identified to be the cause. Research by Dongsheng and associates in 2005 has contributed to the theory that obesity is a trigger for onset of Type II DM. Their research demonstrated that obesity "causes low-grade inflammation which then causes changes in liver function accompanied by hyperglycemia and insulin resistance, a decreased sensitivity to insulin at the tissue level" (Dongsheng et al, 2005 as cited in Timby & Smith, 2013, p. 785). Normally, any time there is inflammation in the body it places the body under stress, so the liver will secrete more glycogen (to turn into glucose) to give the body energy to work at reducing the inflammation. Obesity causes a type of stress on the body which produce an inflammation response which will trigger the liver mechanism to secrete glycogen.
Insulin and glucagon are two hormones that help to regulate the glucose levels in the body. During mealtimes, when glucose level is high, insulin helps to deliver glucose into the cells that need it, while at the same time, lowering the circulating levels. At this time, glucagon levels are decreased. When glucose levels are too low, glucagon sends the signal to the liver to secrete more glycogen to be turned into glucose for energy needs. Unfortunately, in individuals with diabetes, the opposite occurs. While eating, their glucagon levels rise, which causes blood sugar levels to rise after the meal.
Insulin resistance is another reason why glucose levels remain elevated after a meal time. The beta cells produce insulin in response to the elevated glucose levels in the blood but the glucose levels remain higher than normal because there is a deficiency in transmembrane glucose transporters found on the surface of cells. These transporters form channels that help to allow glucose to enter into the cells. People with Type II DM may only have function of these transporters at 20% efficiency, which is why glucose levels rise, causing hyperglycemia. Exercise, an important part of glucose control therapy, allows increased transmembrane glucose transporter levels in the skeletal muscles, therefore reducing circulating blood glucose levels. This is why it is so important to educate and encourage people with diabetes to exercise as part of their lifestyle modification plan.
Over time, the overstimulation of beta cells in the pancreas will cause them to be exhausted and give up, which will result in a decline of insulin production, which can lead the person with Type II DM to become insulin deficient, like in Type I (Timby & Smith, 2013).
From here, the effects are similar to Type I. The glucose cannot enter the cells properly due to insufficient insulin production or resistance so the body starts to breakdown fats for energy, producing ketones. The patient will experience polydipsia, polyuria, polyphagia, and hyperosmolar hyperglycemic nonketotic syndrome (HHNKS) can occur if not treated immediately.
Insulin and glucagon are two hormones that help to regulate the glucose levels in the body. During mealtimes, when glucose level is high, insulin helps to deliver glucose into the cells that need it, while at the same time, lowering the circulating levels. At this time, glucagon levels are decreased. When glucose levels are too low, glucagon sends the signal to the liver to secrete more glycogen to be turned into glucose for energy needs. Unfortunately, in individuals with diabetes, the opposite occurs. While eating, their glucagon levels rise, which causes blood sugar levels to rise after the meal.
Insulin resistance is another reason why glucose levels remain elevated after a meal time. The beta cells produce insulin in response to the elevated glucose levels in the blood but the glucose levels remain higher than normal because there is a deficiency in transmembrane glucose transporters found on the surface of cells. These transporters form channels that help to allow glucose to enter into the cells. People with Type II DM may only have function of these transporters at 20% efficiency, which is why glucose levels rise, causing hyperglycemia. Exercise, an important part of glucose control therapy, allows increased transmembrane glucose transporter levels in the skeletal muscles, therefore reducing circulating blood glucose levels. This is why it is so important to educate and encourage people with diabetes to exercise as part of their lifestyle modification plan.
Over time, the overstimulation of beta cells in the pancreas will cause them to be exhausted and give up, which will result in a decline of insulin production, which can lead the person with Type II DM to become insulin deficient, like in Type I (Timby & Smith, 2013).
From here, the effects are similar to Type I. The glucose cannot enter the cells properly due to insufficient insulin production or resistance so the body starts to breakdown fats for energy, producing ketones. The patient will experience polydipsia, polyuria, polyphagia, and hyperosmolar hyperglycemic nonketotic syndrome (HHNKS) can occur if not treated immediately.